How to Tell If Your Peptides Are Legit in 2025
Table of Contents
-
Introduction: Why Quality Control Matters More Than Ever
-
Step 1: Understanding a Proper COA
-
Step 2: The Importance of Batch Testing
-
Step 3: Multi-Vial Testing and Consistency
-
Step 4: Endotoxin and Sterility Testing — The Final Layer of Safety
-
Conclusion: Research Smart, Research Safe
If you are curious as to what a great Quality Program Is Read Here: From Modern Aminos!
1. Introduction: Why Quality Control Matters More Than Ever
As the peptide industry continues to grow at record speed in 2025, new suppliers are appearing almost daily. While this creates more options for researchers, it also opens the door to low-quality, mislabeled, or contaminated products.
Understanding how to verify whether your peptides are legitimate is absolutely essential. Poorly tested products can lead to inaccurate research results, wasted money, and potentially dangerous outcomes if used irresponsibly. That’s why quality control (QC) — and knowing how to evaluate it — is one of the most important skills any serious researcher can have.
2. Step 1: Understanding a Proper COA
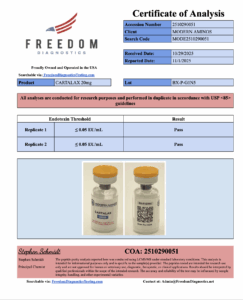
A Certificate of Analysis (COA) is the foundation of any quality peptide company. However, not all COAs are created equal. Many less-reputable sellers will upload generic, outdated, or even falsified certificates.
When reviewing a COA, here’s what to look for:
-
Reputable third-party labs: The testing should come from trusted, independent laboratories such as Vanguard or Chromate Analytical. These labs have transparent methodologies and are known for rigorous standards.
-
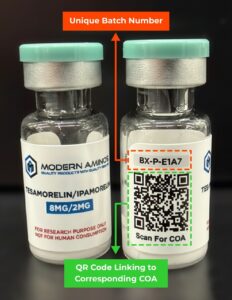
Specific details: A legitimate COA should include the peptide name, batch number, purity percentage, concentration, and analytical methods used (such as HPLC or mass spectrometry).
-
Recent dates: The test date should align with the current product batch, not a generic COA from months or years ago.
When these details align, you can have far more confidence in the accuracy and authenticity of your peptide.
3. Step 2: The Importance of Batch Testing
Batch testing ensures that every single production run of a peptide is verified for purity, concentration, and identity. This allows researchers to match their vial directly to the specific batch tested by the lab — a critical step for transparency and traceability.
If a company doesn’t perform batch testing, there’s no guarantee that what’s in your vial matches the COA they’re advertising. In other words, a generic COA might represent a completely different production run.
Modern Aminos sets an excellent standard for this process, offering clear batch numbers and linked COAs for every peptide. This level of visibility builds trust and accountability that the research community depends on.
4. Step 3: Multi-Vial Testing and Consistency
Even with batch testing, a company should also perform multi-vial testing — meaning multiple vials from the same batch are tested to confirm consistency across production. This reduces the risk of isolated contamination or degradation that might occur during handling or bottling.
Companies that invest in multi-vial testing demonstrate they take their QC process seriously, ensuring the entire batch meets the claimed purity and potency standards.
5. Step 4: Endotoxin and Sterility Testing — The Final Layer of Safety
While optional for many research applications, endotoxin and sterility testing represent an advanced level of safety and quality.
-
Endotoxin Testing: Endotoxins are toxic substances produced by bacteria. Even trace amounts can disrupt biological research or skew results. Testing for endotoxins ensures the compound is clean and free from bacterial contamination.
-
Sterility Testing: This becomes especially important when working with pre-mixed amino blends or reconstituted peptides, where contamination risk is higher. Companies that perform sterility testing show an added commitment to ensuring product safety and purity.

6. Conclusion: Research Smart, Research Safe
With so many peptide suppliers appearing in 2025, understanding how to evaluate testing and quality control isn’t optional — it’s essential.
Always make sure the company you purchase from:
-
Uses reputable third-party labs (like Vanguard or Chromate)
-
Performs multi-vial and batch testing
-
Provides transparent COAs with key details
-
Offers optional endotoxin or sterility testing for added assurance
If you’re looking for a reliable example of proper QC in action, check out Modern Aminos, one of the most trusted names in the research space.


